Cancer is one of the leading causes of death in the United States and accounts for approximately 8 million deaths per year worldwide. Although there is an increasing number of therapeutic options available for patients with cancer, their efficacy is time-limited and non-curative. Approximately 50-60% of cancer patients in the United States utilize agents derived from different parts of plants or nutrients (complementary and alternative medicine), exclusively or concurrently with traditional therapeutic regime such as chemotherapy and/or radiation therapy. The need for new drugs has prompted studies evaluating possible Anti-Cancer agents in fruits, vegetables, herbs and spices. Saffron, a spice and a food colorant present in the dry stigmas of the plant Crocus Sativus L., has been used as an herbal remedy for various ailments including cancer by the ancient Arabian, Indian and Chinese cultures. Crocetin, an important carotenoid constituent of saffron, has shown significant potential as an anti-tumor agent in animal models and cell culture systems. Crocetin affects the growth of cancer cells by inhibiting nucleic acid synthesis, enhancing anti-oxidative system, inducing apoptosis and hindering growth factor signaling pathways. This review discusses the studies on cancer preventive potential of Crocetin and its future use as an anticancer agent.
Pharmacological Properties of Crocetin
Animal studies indicated that the oral LD50 of crocetin or saffron was 20.7 g/kg body weight when administered as decoction. It has been demonstrated that oral administration of saffron extract at doses from 0.1 to 5 g/kg was non-toxic in mice. The stability of crocetin is also dependent upon temperature, light and humidity on degradation of potency under storage conditions. Ingredients of saffron including crocetin can be stored under −20°C. Its pharmacological activity remains unaltered for at least 2 years.
Saffron and its constituents have been demonstrated significant medical-biological activities as described by Abdullaev and Espinosa-Aguirre . It has been reported that saffron derived compounds like crocetin showed significant effects on coronary heart disease by inhibiting lipoprotein oxidation, on neurodegenerative diseases, on retinal function recovery by increasing retinal blood flow, on reducing blood pressure and maintaining kidney function and also used as anticonvulsive agent. Most notable discovery in relation to crocetin is to enhance oxygen diffusivity during shock and reperfusion that ultimately lead to increased ATP production.
.jpg)
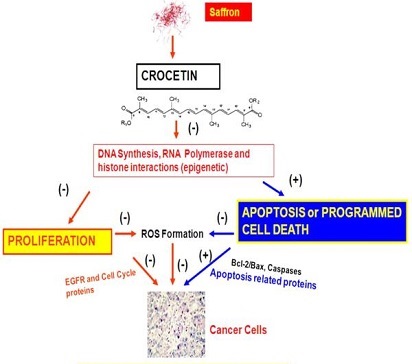
Although there are many reports on the effects of saffron extract on cancer, there is very limited information available for crocetin effects on cancer. Saffron and its derivatives particularly crocetin have demonstrated significant anticancer activity in breast, lung, pancreatic and leukemic cells. summarizes the effects of crocetin against several cancer types and also presents the underlying mechanisms of action.
Crocetin and its analogues separated from different Crocus species inhibited breast cancer cell proliferation. MCF-7 and MDA-MB-231 breast cancer cells showed concentration-dependent inhibition of proliferation by crocetin and this effect was independent of estrogen receptor. This study also suggested that crocetin can be used as chemopreventive agent in breast cancer. Crocetin also exerts proapoptotic effect in MCF-7 breast cancer cells indicating caspase dependent pathway through increased expression of Bax protein.
It has been reported that HeLa cells demonstrated significant reduction of colony formation and cellular RNA and DNA synthesis by crocetin like compounds from saffron. Saffron was found to decrease the viability of HeLa cells. In that study, crocetin showed dose-dependent inhibition of DNA, RNA and protein synthesis (over the range of 1-200 μg/ml). Crocetin also suppressed DNA-dependent RNA polymerase II enzyme following inhibition of RNA synthesis. Some of the similar derivatives of crocetin derived from saffron, such as crocin, safranal and picrocrocin, showed significant reduction of growth of HeLa cells and exhibited wide cytoplasmic vacuole-like areas, reduced cytoplasm, cell shrinkage and pyknotic nuclei suggesting apoptosis. It has been also demonstrated using UV-spectroscopy that crocetin interacts with tRNA internally with a binding constant 1.4 ± 0.31 μM. This indicated the binding activity of crocetin at molecular level suggesting its cancer preventive effect.
In another study, crocin (obtained from Crocus sativas) significantly inhibited the growth of colorectal cancer cells and it has been suggested as a viable agent for the treatment of colorectal cancer.
In two other studies using promyelocytic leukemia (HL60) and human myelogenous leukemia (K562) cells, crocetin demonstrated significant cytoxicity and inhibited proliferation with as low as 0.8 μM concentrations. Cytotoxicity of crocetin on various other leukemic cell lines (L1210 and P388) has also been reported.
The cytotoxicity and DNA-adduct formation of rat liver microsomes activated by aflatoxin B1(AFB1) in the C3H10T1/2 fibroblast cells are significantly inhibited by pretreatment of crocetin. Crocetin treatment resulted in a decrease in AFB1-DNA adduct formation in vitro that suggested the protective effect of crocetin on the AFB1-cytotoxity due to the elevation of cytosolic glutathione (GSH) following the activities of GSH-S-transferase (GST) formation as cellular defense mechanisms. Crocetin pretreatment in rats protected hepatic AFB1-induced hepatic damage and AFB1-DNA adducts formation due to the elevation hepatic GSH, activities of GST and glutathione peroxidase (GSH-Px). In another study, significant suppression of AFB1-induced hepatotoxic lesions was observed as indicated by reduction of activities of serum aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase and gamma-glutamyl transpeptidase (GGT) by crocetin in rats. Inhibitory effect of crocetin on benzo[a]pyrene-induced genotoxicity and neoplastic transformation in C3H1OT1/2 cells is due to a mechanism that increased the activity of GSH and reduced the formation of benzo[a]pyrene-DNA adducts. Crocetin also inhibited the formation of malondialdehyde (MDA), a marker for lipid peroxidation, induced by reactive oxygen species (ROS) generated by the activity of xanthine oxidase (XO) in primary hepatocytes and protected against oxidative damage. Therefore, these studies indicated that crocetin displayed protective action against the ROS due to direct scavenging that inhibited free radical production following neoplastic transformation. Saffron was also reported to decrease the viability of HepG2 cells.
Another in vivo study reported that crocetin has antitumor activity in a lung cancer animal model by scavenging free radicals and increasing the activity of drug metabolizing enzymes. Crocetin scavenged free radicals as evidenced by inhibiting lipid peroxidation and increase of the activity of GST, GSH-Px, catalase, and superoxide dismutase due to crocetin treatment. Crocetin also decreased marker enzymes such as arylhydrocarbon-hydroxylase (AHH), lactate dehydrogenase (LDH), GGT, adenosine deaminase (ADA) and 5′-nucleotidase related to carcinogen following administration of benzo[a]pyrene (B[a]P) in lung tissues. Furthermore, Magesh et al. also demonstrated that crocetin was capable of inhibiting proliferation of lung cancer cells as determined by PCNA, glycoproteins and polyamine synthesis. This study strongly suggested that the protective effect of crocetin on B[a]P-induced lung carcinogenesis in Swiss albino mice are likely due to the inhibitory effects on polyamine synthesis and glycoprotein alterations. Crocetin has shown inhibitory effects on intracellular nucleic acid synthesis and colony formation of A549 (lung carcinoma) and VA13 (SV-40 transformed fetal lung fibroblast) cells.
Our group has studied, for the first time, the anticancer potential of crocetin in pancreatic cancer by using various pancreatic cancer cells as well as a xenograft athymic mouse model. In this study, crocetin inhibited the proliferation of Mia PaCa2, BxPC3, Capan-1 and ASCPC-1 cells. Crocetin significantly inhibited cell distribution in S-phase impairing DNA replication. This confirms the inhibition of DNA synthesis in crocetin-treated pancreatic cancer cells. The increase of accumulation of cells indicated G2/M arrest in Mia-PaCa-2 cells due to crocetin treatment. The cell cycle entry of cells depends upon the activity of several regulatory proteins including Cdc-2, Cdc-25c, cyclin B1 as well as other proteins. The expression of cyclin B1 was assessed and the inhibition of expression of cyclin B1 and inactivation of Cdc2 due to inhibition of Cdc-25c could be the major step for arrest in G2/M phase following crocetin treatment. Overall, these studies evaluating major cell cycle checkpoint proteins indicate crocetin is effective as antiproliferative agent in accomplishing cell cycle arrest. Crocetin significantly reduced epidermal growth factor receptor (EGFR) expression and phosphorylation as indicated by the ratio of phosphorylated and unphosphorylated EGFR proteins in MIA-PaCA-2 cells. Crocetin showed antitumorigenic effects on pancreatic cancer in an athymic (nude) mice model by regressing tumor growth following inhibition of proliferation as determined by PCNA and EGFR expression in crocetin-treated animals. Immunohistochemical and Western blot analysis revealed a significant decrease of EGFR phosphorylation and expression in the tumors developed in nude mice after crocetin treatment. This indicated that crocetin is an effective inhibitor of EGFR activity that in turn impaired growth of pancreatic cancer. In this study, the ratio of Bax/Bcl-2 was significantly increased both Mia PaCa-2 cells and in pancreatic tumors after crocetin treatment, which suggested that the reduction on imbalance between antiapoptotic (Bcl-2) and proapoptotic (Bax) proteins could be a major factor for the antitumorigenic activity of crocetin. It has been demonstrated recently that crocetin in combination with lower doses of paclitaxol or cisplatin inhibited proliferation and stimulated apoptosis in pancreatic cancer (unpublished observations). This study indicated that crocetin showed promising effect in combinational therapy with conventional chemotherapeutic agents.
At present, it is not known that whether the effect of crocetin on pancreatic cancer regression is receptor dependent or independent mechanisms, which is being investigated in our laboratory. Nevertheless, based on currently available data, it can be concluded that crocetin could be used as a novel therapy for pancreatic cancer due to significant antitumorigenic effect and thereby can be of great therapeutic benefit against this aggressive cancer.
Administration of crocetin delayed the onset of skin tumor initiation and decreased its tumor formation initiated with dimethylbenz anthracene (DMBA) and promoted by croton oil in Swiss-Webster mice. Similar antitumor activity of crocetin also observed in hairless mice with skin tumors developed by the application of DMBA and croton oil.
In summary, crocetin demonstrated significant inhibitory effect on the growth of a number of cancer cells. The tumor growth-suppressing effect of crocetin could be due to the reduction in the synthesis of DNA, RNA and protein by crocetin in tumor cells. It has also been demonstrated that crocetin inhibited RNA polymerase II activity in neoplastic cells. Crocetin also interferes with histone H1 structure and H1-DNA interaction suggesting that epigenetic mechanisms could be implicated in the anticarcinogenic action of this natural product .
.jpg)
The exact mechanism of protective activity of crocetin is not clear at present but several hypotheses have been put forwarded which suggest that carotenoids can be converted to vitamin A, can enhance carcinogen metabolism, can act as antioxidant or inhibit nucleic acid synthesis. summarizes the possible mechanisms of action of crocetin. We propose that crocetin inhibits DNA synthesis and RNA polymerase II activity, that in turn, inhibits proliferation by impairing EGFR, phospho Cdc-2, Cdc25c, cyclin B1 and increases apoptosis by enhancing the ratio of Bax/Bcl-2 that ultimately inhibits tumor formation. Crocetin also inhibits oxidant injury due to lipid peroxidation and these antioxidant effects could be responsible for inhibition of tumor formation. All these studies warrant more research to evaluate the exact antitumor mechanism(s) of crocetin before embarking on clinical trials. Nevertheless, the emerging in vitro and in vivo studies on several types of cancers as presented in this review underscore the potential of crocetin in the prevention and treatment of human malignancies.
Crocetin belongs to the large family of natural dyes known as carotenoids, but it does not have a pro-vitamin function. The constituents of this class of small molecule compounds are mostly polyunsaturated hydrocarbons (the formula is C40H56) or their oxygenated derivatives. There are small groups of caretonoids that are carboxylic acids. Among those groups there is crocetin (the glycan of crocetin), 8,8′diapo-8,8′-carotenic acid, characterized by a diterpenic and symmetrical structure with alternating double bonds and four methyl groups. The chain is stabilized in the terminal parts by two carboxylic groups. Its elementary composition is C20H24O4 and its molecular weight is 328.4. It is slightly soluble in aqueous solution (20 μM at pH 8.0) and it is soluble in organic bases, such as, pyridine. Various analytical methods have been developed including thin-layer chromatography. Chemical analyses of dry stigma of saffron extracts have revealed that characteristic compounds of saffron include the carotenoids, namely crocin and crocetin and the monoterpene aldehydes picocrocin and safranal. Crocetin is an amphiphilic low molecular weight carotenoid compound and consists of a C-20 carbon chain with multiple double bonds, and a carboxylic acid group at each end of the molecule. The structure of crocetin is presented in, where R1 and R2 = H.
William G. Gutheil,a Gregory Reed,b Amitabha Ray,c and Animesh Dhard,e,*